About Us
Glaukos is transforming the treatment of chronic eye diseases with novel therapies that provide sustainable solutions to important clinical needs.

Who We Are
Glaukos is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal disease.
Our Mission
Innovation is at the core of everything we do. Our mission at Glaukos is to truly transform vision by pioneering novel, dropless platforms that can meaningfully advance the standard of care and improve outcomes for patients suffering from sight-threatening chronic eye diseases. Our mantra “We’ll Go First” embodies our commitment and determination to take chances and push the limits of science and technology to disrupt and improve the legacy treatment paradigms in glaucoma, corneal disorders and retinal diseases.
Glaukos Corporate Overview
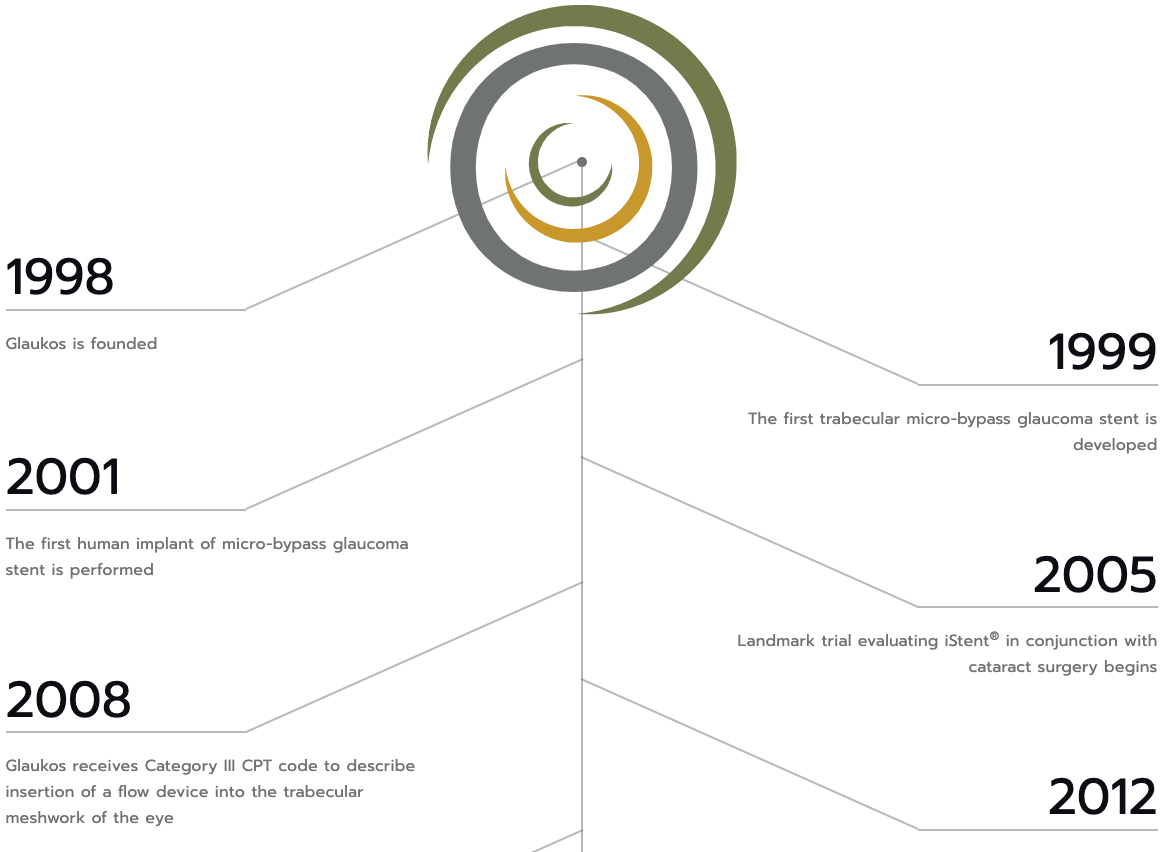
 Founded in 1998, Glaukos is an ophthalmic medical technology and pharmaceutical company focused on developing and commercializing novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases.
Founded in 1998, Glaukos is an ophthalmic medical technology and pharmaceutical company focused on developing and commercializing novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases.
Glaukos first developed Micro-Invasive Glaucoma Surgery (MIGS) as an alternative to the traditional glaucoma treatment paradigm, launching its first MIGS device commercially in 2012, and continues to develop a portfolio of technologically distinct and leverageable platforms to support ongoing pharmaceutical and medical device innovations. Products or product candidates for each of these platforms are designed to advance the standard of care through better treatment options across the areas of glaucoma, corneal disorders and retinal diseases.
Our company completed an initial public offering in June of 2015, and our shares are traded on the New York Stock Exchange under the ticker symbol “GKOS”. Our global headquarters is located in Aliso Viejo, California, and we have additional locations in the United States, Canada, United Kingdom, Germany, Japan, Australia and Brazil.

Code of Conduct
This reflects our core values of integrity, transparency, and accountability—principles that guide both our internal operations and our engagement with the broader community.

Prioritizing Corporate
Responsibility
We’re proud of the corporate citizenship efforts of the Glaukos Charitable Foundation, Company and employees, and pledge to continue this important work in helping improve the lives of those suffering and in need.
Careers at Glaukos
Help put the next wave of disruptive technologies on the market.
Work with a team that’s dedicated to making a difference in the lives of patients worldwide.