IMPORTANT SAFETY INFORMATION
The most common side effects were red eye, haze, sensitivity to light, disruption of surface cells of the cornea, eye pain, eye irritation, watery eyes, swelling of eyelid, fine white lines in the cornea, reduced sharpness of vision, dry eye, and eye inflammation.
You should not have the EPIOXA™ corneal collagen cross-linking procedure if you have a known hypersensitivity to any ingredients in the product, have had cataract surgery and either did not receive an artificial lens in your eye or received a non-UV blocking artificial lens, have a history of herpetic keratitis, or are pregnant.
If you have any additional questions, please contact your doctor. Please see full Prescribing Information for EPIOXA HD and EPIOXA.
You are encouraged to report all side effects to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088. You may also call Glaukos at 1-888-404-1644.
APPROVED USES
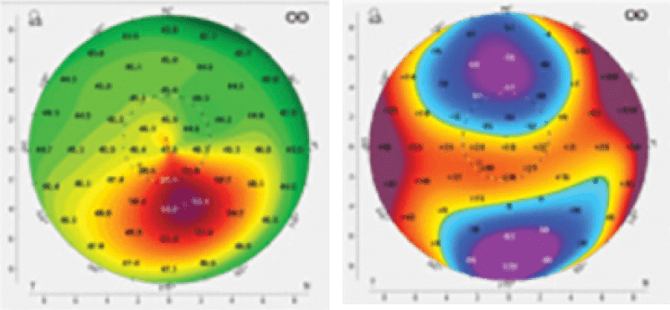
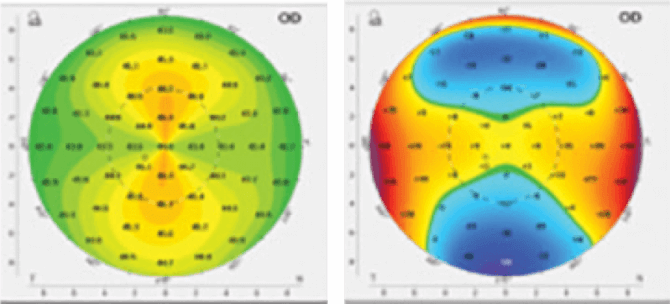
The EPIOXA corneal collagen cross-linking procedure is performed by your eye doctor for the treatment of keratoconus. It does not require the removal of the corneal epithelium (outermost layer of the front of the eye). It consists of EPIOXA™ HD (riboflavin 5′-phosphate ophthalmic solution) 0.239% and EPIOXA™ (riboflavin 5′-phosphate ophthalmic solution) 0.177%, which are prescription eye drops used in combination with the O2n™ System and Boost Goggles®.
The EPIOXA corneal collagen cross-linking procedure is for the treatment of keratoconus in adults and pediatric patients 13 years of age and older.