Corneal Health Solutions
Get There in Time
We’re transforming the standard of care for patients with progressive keratoconus and other corneal ectatic conditions through our commitment to addressing important unmet clinical needs in corneal health.
With inspired innovation, a customer-centric focus, and prolific market access capabilities, we are in the constant pursuit of developing proven solutions in corneal health that empower eye care professionals to deliver optimal care for patients
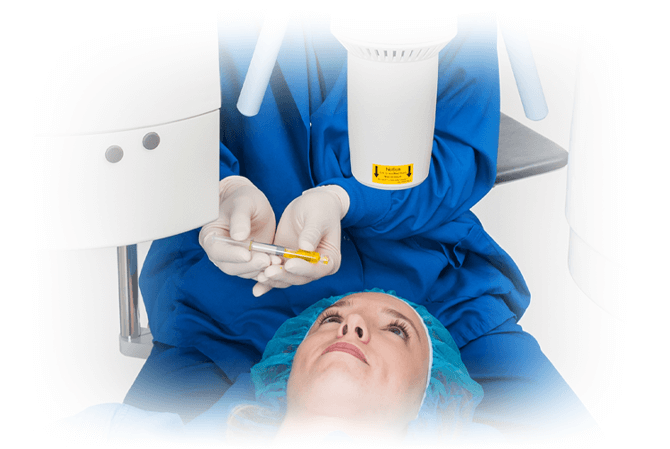
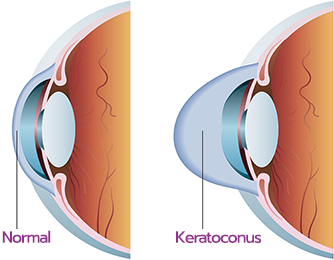
What are the signs and symptoms of progressive keratoconus, and how can corneal cross-linking help?
Contact Us Today
Explore additional information about Corneal treatment
Request
More Info
"*" indicates required fields
